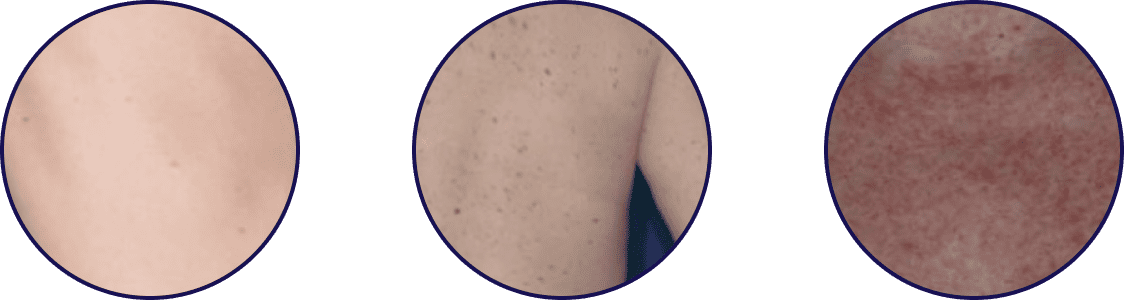
Symptoms of systemic mastocytosis (SM) may be mistaken as other disorders and conditions, leading to diagnostic delays1,2
It is important to recognize the symptoms of SM to help reduce the time to diagnosis and treatment
Historically, it has taken ~6 years for patients living with SM to receive a diagnosis and often involves a diverse group of specialists.1,3 Symptoms of SM include skin lesions, diarrhea, and anaphylaxis, and can be perceived as a variety of related disorders.2,4
Expand to see how these disorders are different from SM